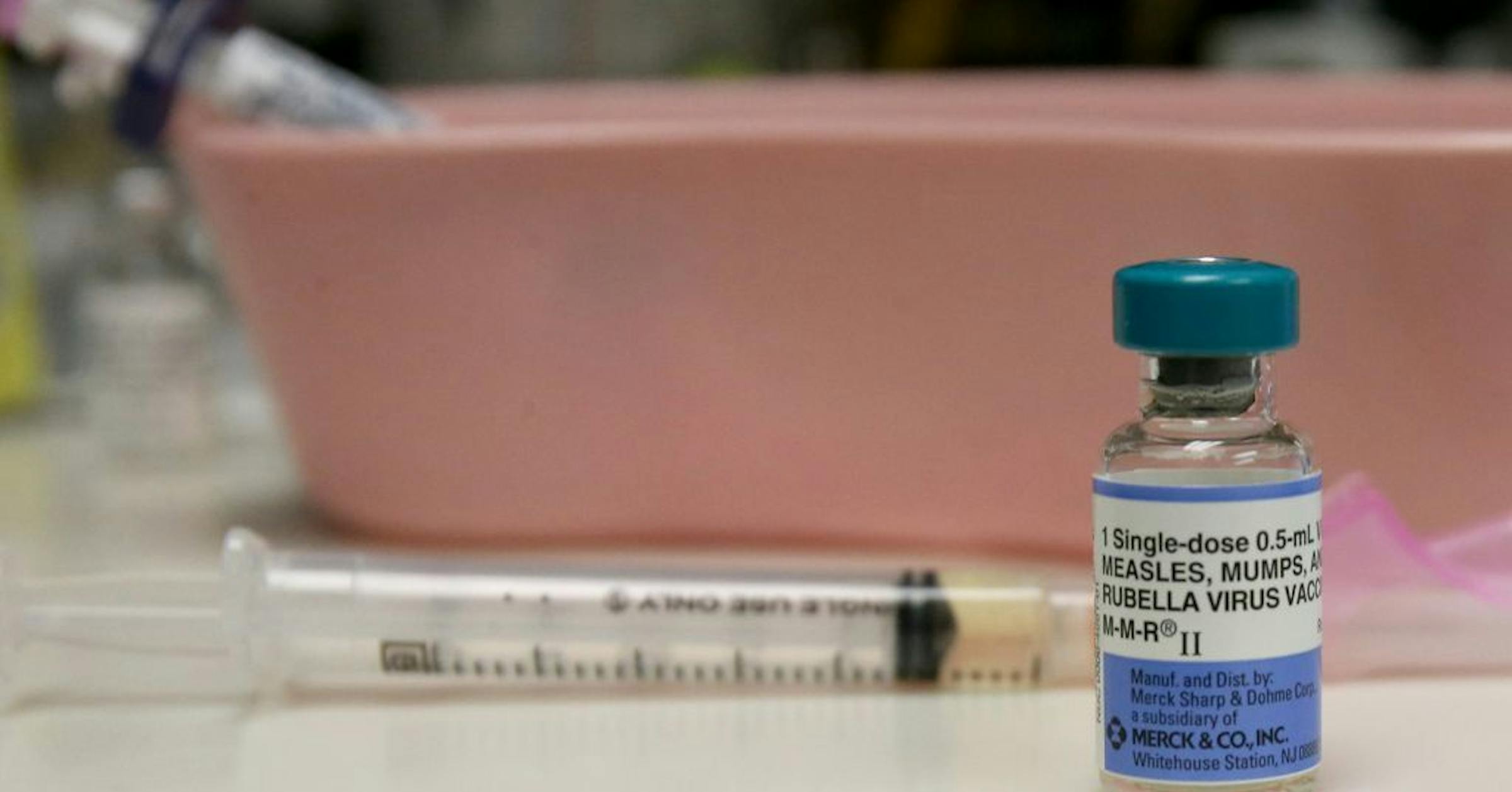
FDA Approves New Generic Mifepristone, Expanding Access to Medication Abortion

Expanding Access to Medication Abortion
The FDA’s recent approval of a new generic form of mifepristone—manufactured by Evita Solutions—marks a pivotal moment in reproductive healthcare access. This 200-mg oral tablet, designed to terminate pregnancies up to 10 weeks, joins two existing generics, broadening options for patients as state-level restrictions intensify[1]. Mifepristone, used alongside misoprostol, is a cornerstone of medication abortion, accounting for roughly two-thirds of such procedures in the U.S.[1] The approval comes as the national debate over abortion rights remains deeply polarized, with advocates highlighting increased affordability and availability, while opponents voice strong objections to any expansion of abortion access.
Political and Practical Implications
While the FDA’s decision is framed by supporters as a step toward equitable healthcare, it has sparked immediate backlash from conservative groups, who argue it undermines efforts to limit abortion nationwide. The introduction of a third generic manufacturer could help stabilize supply chains in regions where access is threatened by legal challenges or shortages[1]. For healthcare providers, this development offers more flexibility in sourcing medications, potentially reducing costs for patients. Yet, the ongoing political and legal battles ensure that the availability of mifepristone will remain a flashpoint in the broader reproductive rights conversation for the foreseeable future.
About the Organizations Mentioned
FDA
## Overview The **U.S. Food and Drug Administration (FDA)** is a federal agency within the Department of Health and Human Services responsible for protecting public health by ensuring the safety, efficacy, and security of a wide range of products, including human and veterinary drugs, biologics, medical devices, food, cosmetics, and products that emit radiation[1][2][3]. Its mission is to advance public health by helping to speed innovations that make medical products safer, more effective, and more affordable, while providing the public with accurate, science-based information about these products[1]. ## Functions and Regulatory Scope The FDA’s regulatory authority is expansive. It oversees the approval, manufacturing, marketing, and distribution of prescription and over-the-counter drugs, vaccines, blood products, medical devices (from simple tongue depressors to complex pacemakers), dietary supplements, most foods (except some meat, poultry, and egg products regulated by the USDA), cosmetics, and tobacco products[1][2][5]. The agency also regulates electronic products that emit radiation, such as X-ray machines and microwave ovens[2][5]. Importantly, the FDA does not regulate the practice of medicine, medical services, product pricing, or health insurance reimbursement[2]. The FDA achieves its goals through a combination of **premarket reviews**, **post-market surveillance**, **facility inspections**, **enforcement actions**, and **public education**[3][4]. It maintains several adverse event reporting systems—such as MedWatch and VAERS—to monitor product safety after they reach the market[4]. The agency also plays a key role in the nation’s counterterrorism efforts by ensuring food supply security and fostering the development of medical countermeasures[1]. ## History and Key Achievements Established in 1906 with the passage of the Pure Food and Drugs Act, the FDA’s origins trace back to efforts to combat adulterated and misbranded food and drugs. Its regulatory powers expanded significantly with the
Evita Solutions
Evita Solutions refers to multiple distinct organizations across various industries, but the most prominent current entity under this name is **Evita Solutions LLC**, a pharmaceutical company specializing in healthcare solutions, notably in reproductive health. In September 2025, Evita Solutions LLC received FDA approval for its generic version of mifepristone, a medication used for terminating early pregnancies up to 70 days from the last menstrual period. This approval marks a significant milestone as Evita Solutions became the second company to offer a generic mifepristone in the United States, aiming to improve access to safe, affordable, and compassionate abortion care amid ongoing political and legal challenges surrounding reproductive rights[6][7]. Evita Solutions LLC emphasizes compliance with the FDA's Risk Evaluation and Mitigation Strategy (REMS), requiring certified prescribers and specific healthcare settings to dispense the drug, ensuring patient safety. The company advocates for broad access to high-quality abortion medication while navigating a complex regulatory landscape shaped by state-level restrictions and lawsuits[6][7]. Other organizations bearing the name "Evita Solutions" exist but are unrelated to this pharmaceutical company. For example, a UK-based company named EVITA SOLUTIONS LTD was a private limited company involved in machinery manufacturing but was dissolved in 2014[2]. Additionally, there are IT and software firms in India named Evita Soft Solutions focused on IT services and software development[4][5], and Envita Solutions in North America specializing in innovative waste management and sustainability technologies[1]. These entities operate in entirely different sectors and have no direct connection with Evita Solutions LLC. In summary, the most newsworthy and current "Evita Solutions" is the pharmaceutical company advancing reproductive healthcare through FDA-approved generic medications. Its recent FDA approval in 2025 positions it as a significant player in the medication abortion market, emphasizing accessibility and safety in a politically sensitive arena[6][7].