CDC Revises Child Vaccine Schedule Toward Individualized Hepatitis B Birth Dose

CDC Revises Child Vaccine Schedule
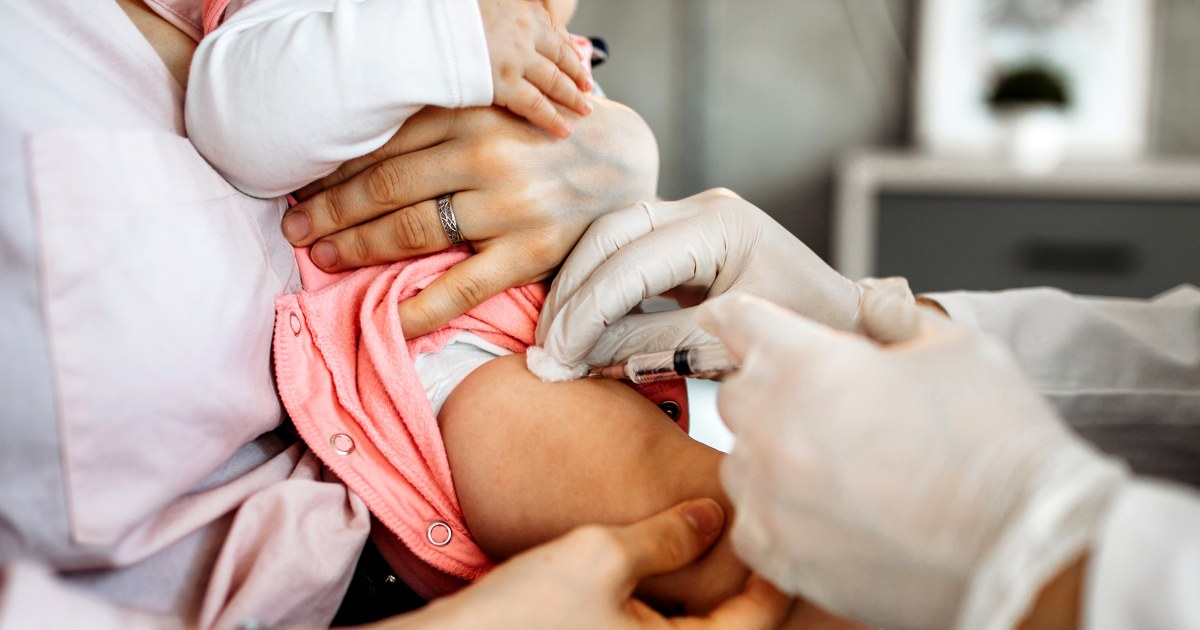
The CDC’s Advisory Committee on Immunization Practices (ACIP) has made its most significant change to the childhood vaccine schedule in decades by removing the universal recommendation for newborns to receive the hepatitis B vaccine at birth. Instead, the hepatitis B birth dose will now be recommended only if the mother tests positive or her status is unknown, shifting the decision to parents and healthcare providers for other cases.
New Approach to Vaccination Decisions
This change reflects a broader move toward individualized vaccine decisions, emphasizing informed consent and shared decision-making rather than blanket mandates. The committee also discussed potentially adjusting the total number of hepatitis B doses based on antibody testing, which could reduce unnecessary vaccinations for some infants.
Implications for Parents and Providers
While some experts express concern about deviating from established schedules, the update aims to balance vaccine benefits with parental choice and the latest safety data. This revision may signal further changes in childhood immunization practices under the current CDC leadership.
About the Organizations Mentioned
Centers for Disease Control and Prevention
The Centers for Disease Control and Prevention (CDC) is a premier U.S. public health agency established on July 1, 1946, originally as the Communicable Disease Center. It evolved from the wartime Malaria Control in War Areas program (MCWA) created during World War II to combat malaria around military bases in the southern United States[1][3][7]. Headquartered in Atlanta, Georgia, the CDC has grown from a regional malaria control unit to a comprehensive national and global health protection agency. The CDC’s mission is to protect public health and safety through disease control and prevention, health promotion, and emergency preparedness. It investigates and responds to emerging health threats such as infectious diseases—including COVID-19, influenza, and bioterrorism agents—as well as chronic diseases, injuries, workplace hazards, environmental health threats, and more[2][6]. The agency conducts scientific research via over 200 specialized laboratories nationwide, supports public health workforce development, and communicates critical health information to the public[6][5]. Throughout its history, the CDC has expanded its scope and structure significantly. It was renamed the Center for Disease Control in 1970, then the Centers for Disease Control in 1980 as it incorporated multiple centers, and finally adopted the current name, Centers for Disease Control and Prevention, in 1992 to emphasize prevention efforts while retaining the CDC acronym for recognition[2][8]. Its organizational breadth now includes centers focused on infectious diseases, chronic diseases, environmental health, injury prevention, occupational safety, and health statistics. Notable achievements include leading vaccination campaigns against diseases like measles and rubella, advancing injury prevention, and mounting global efforts against infectious outbreaks. The CDC also played a pivotal role in combating antibiotic misuse and bioterrorism preparedness. Despite past controversies like the Tuskegee syphilis study, the agency remains a leader in epidemiology and public health innovation, employing a multidisciplinary workforce of scientists, clinicians, and public health experts dedicated t
Advisory Committee on Immunization Practices
## Advisory Committee on Immunization Practices (ACIP): Overview, History, and Impact The **Advisory Committee on Immunization Practices (ACIP)** is a federal advisory committee under the U.S. Centers for Disease Control and Prevention (CDC) responsible for providing expert recommendations on the use of vaccines and related agents to control vaccine-preventable diseases in the U.S. civilian population[1][2][6]. Its guidance covers routine immunization schedules for children and adults, as well as nonroutine situations such as outbreaks and travel[2]. ACIP’s recommendations are foundational for both public health policy and clinical practice, influencing everything from school entry requirements to insurance coverage for vaccines[1]. ## History and Structure Established in March 1964 by the U.S. Surgeon General, ACIP was created under Section 222 of the Public Health Service Act to provide ongoing, independent advice to the Secretary of Health and Human Services (HHS) on vaccine policy[1][2]. The committee comprises medical and public health experts, including both voting and liaison members, and operates through regular public meetings and specialized work groups that review evidence and draft recommendations[2][6]. Final recommendations require a majority vote and are published in CDC’s Morbidity and Mortality Weekly Report (MMWR), making them official federal guidance[1][2]. ## Key Achievements ACIP’s recommendations have shaped the U.S. immunization landscape for over half a century, ensuring the integration of new vaccines (such as those for HPV, hepatitis B, and COVID-19) into routine care as soon as they are licensed[1][2]. During the COVID-19 pandemic, ACIP played a pivotal role in rapidly developing and updating vaccination guidelines, including recommendations for multiple age groups and immunocompromised individuals[5]. The committee’s rigorous, evidence-based process is designed to balance scientific rigor with public transparency, often assessing new vaccines in parallel with FDA approval[7]. ## Current Status